How the FDA Oversees Foreign Drug Factories Making Your Medicines
Most of the generic pills you take - from blood pressure meds to antibiotics - aren’t made in the U.S. They’re made overseas, mostly in India and China. And yet, the FDA still has to make sure they’re safe, pure, and work just like the brand-name versions. That’s not a small task. In 2025, about 80% of the active ingredients in U.S. drugs came from foreign factories. That means millions of Americans rely on drugmakers halfway across the world to follow the same strict rules as companies in Ohio or Pennsylvania. But for years, the system wasn’t fair.
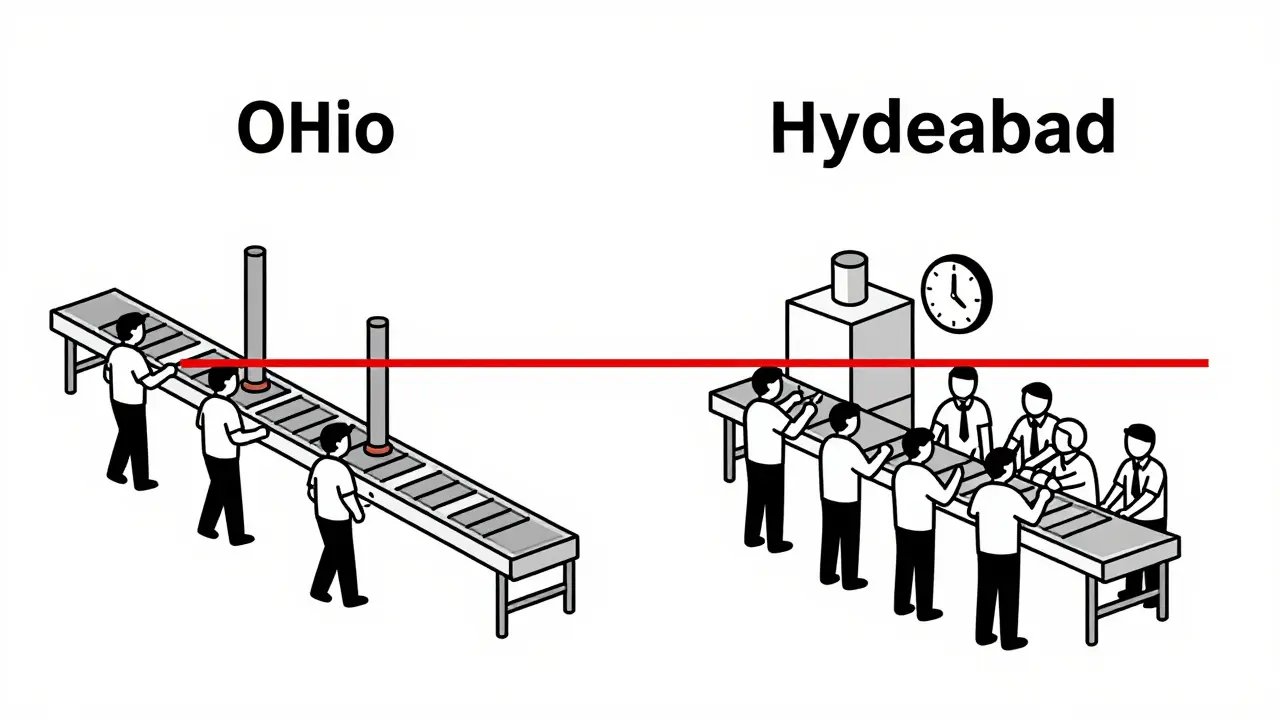
The Double Standard in Inspections
Before May 2025, there was a clear gap in how the FDA checked on drug factories. If you ran a generic drug plant in Indiana, inspectors could show up unannounced - no warning, no time to clean up. But if your factory was in Hyderabad or Shanghai, you got 8 to 12 weeks’ notice. That gave companies time to fix problems, hide records, or even temporarily hire extra staff just for the inspection. It wasn’t real oversight. It was a performance.
The numbers told the story. In 2024, foreign facilities had 38.7% of inspections uncovering data integrity issues - like falsified test results or missing logs. Domestic facilities? Only 17.2%. Contamination problems were more than twice as common overseas. The FDA itself admitted this wasn’t working. The system was built on trust, not verification.
What Changed in 2025?
On May 6, 2025, everything shifted. FDA Commissioner Marty Makary announced a new policy: unannounced inspections would jump from 15% to at least 50% of all foreign inspections by mid-2026. This wasn’t just a tweak. It was a full reset. The goal? To catch bad actors before they shipped dangerous drugs to U.S. pharmacies.
Why now? Pressure from Congress, public reports, and real-world failures. ProPublica found that even after the FDA banned a Sun Pharma facility in India in 2021 for serious violations, four of its drugs were still approved for sale in the U.S. That’s not an accident - it’s a system failure. Senators Gillibrand and Scott demanded answers. President Trump signed Executive Order 14135, ordering the FDA to eliminate the inspection gap within 18 months. The clock started ticking.
What the FDA Actually Checks
When an FDA inspector walks into a foreign factory, they’re not looking for clean floors or shiny machines. They’re looking for proof that every step of production is controlled. Here’s what they dig into:
- Documentation: Every batch must have complete records - who made it, when, what tests were run, and what the results were. No backdated entries. No erased data.
- Testing methods: Are the lab tests accurate? Are they validated? Or are they just guessing?
- Contamination control: Are there dust, microbes, or chemical cross-contaminations? A single mistake here can poison thousands of pills.
- Equipment maintenance: Are machines calibrated? Are filters changed? Or are they running on borrowed time?
- Personnel training: Are workers trained properly? Or are they rushed through a 2-hour video and sent to the floor?
They use Form 483 to list every issue. If it’s serious enough, they issue a warning letter. If the company doesn’t fix it? The FDA can block all shipments from that facility. No more drugs. No exceptions.
How Foreign Factories Are Adapting
Companies in India and China are scrambling to keep up. A PDA survey in early 2025 found that 68% of foreign manufacturers expect compliance costs to rise 15-25%. Smaller factories are feeling the squeeze - they don’t have the teams or budgets to run 24/7 quality audits.
But they’re adapting. According to the Generic Pharmaceutical Association, 42% of member companies have already added extra quality checks. Experts recommend:
- Doing monthly mock inspections - pretend the FDA is coming, and act like it’s real.
- Training staff daily on documentation rules. No more shortcuts.
- Keeping real-time digital logs that can’t be altered after the fact.
- Reviewing every batch before it ships - not just for quality, but for paperwork.
It takes 6 to 9 months to fully adjust. But there’s no going back. The days of warning letters and grace periods are over.
The Bigger Picture: Supply Chains and Safety
Over 90% of U.S. generic prescriptions come from foreign manufacturers. India alone supplies 40% of active ingredients. China adds another 13%. That’s not just a business trend - it’s a public health dependency. If one major factory fails, hundreds of thousands of people could go without their meds.
The FDA is trying to reduce that risk. They’re hiring 200 new inspectors by 2026. They’re sharing inspection data with the EU, Japan, and Australia through mutual recognition agreements. And they’re pushing for a system similar to Europe’s - where a Qualified Person in the U.S. must personally certify each batch before it enters the country.
That’s not just theory. Brookings Institution says adopting this model could cut foreign violations by 30-40%. It adds a human layer of accountability. Not just paperwork. Not just a factory manager. A certified professional who bets their career on the safety of every pill.
What This Means for You
You might not think about where your pills come from. But you should. The FDA’s new rules mean better quality control. Fewer contaminated batches. Less risk of fake or weak drugs hitting pharmacy shelves. That’s good news.
But it also means delays. Evaluate Pharma warns that in the short term, 15-20% of generic drug supplies could be disrupted as factories scramble to meet new standards. You might see temporary shortages. You might have to switch brands. That’s not a failure - it’s a correction. The system was broken. Now it’s being fixed.
What Comes Next?
The FDA’s goal is simple: treat every drug factory - whether in Alabama or Ahmedabad - the same. No special treatment. No advance notice. No exceptions.
By 2027, we should see a major drop in violations. Foreign manufacturers who adapt will thrive. Those who resist? They’ll be shut out. The U.S. market doesn’t need cheap drugs - it needs safe ones.
And that’s the bottom line: your medicine should be as safe as if it was made next door. The FDA is finally making sure it is.


Comments (12)